Prepare the onion extract as follows:
1. Add the sodium chloride to the washing-up liquid. Make up to 100 cm3 with distilled water.
2. Cut the onion into small pieces roughly 5
mm square. Place the pieces in a beaker, then
pour on the detergent
/ salt solution.
3. Stir the mixture and maintain it at 60oC
in a water bath for exactly 15
minutes. This
treatment causes the onion cell membranes
to break down. The detergent forms complexes
surrounding the membrane phospholipids
and proteins, causing them to precipitate out of
solution. In addition, sodium
ions from the salt shield the negatively charged phosphate
groups of the DNA molecules causing
them to coalesce. At 60oC, DNAse
enzymes, which
would otherwise start to cut the DNA
into fragments, are partially denatured.
4. Cool the mixture in an ice water
bath for 5 minutes, stirring frequently.
This slows the
breakdown of DNA which would occur
if a high temperature was maintained.
5. Pour the mixture into a liquidizer
and blend for only 5 seconds on high speed.
This degrades
the cell walls and membranes further,
permitting the release of DNA. Do not blend for too
long as this will break up the
DNA fibres.
6. Filter the mixture into the second beaker. Ensure
that the foam on the surface of the liquid
does not contaminate the filtrate.
The filtrate contains soluble proteins and DNA.
NOTE:
The filtrate may be prepared before a lesson and stored in a refrigerator
for 1~2 days if desired.
Separating DNA from the onion extract:
1. Add 2-3 drops of enzyme
to the onion tissue extract in a boiling tube and mix well. The
enzyme will degrade the proteins
associated with the DNA.
2. Form a layer of ice cold ethanol
on top of the onion extract / enzyme mixture
by pouring it
slowly down the side of the boiling
tube. Leave the tube for 2~3 minutes without
disturbing
it. DNA is insoluble in ice-cold ethanol.
Bubbles will form and whilst other compounds in
the mixture dissolve, the DNA
will precipitate into the upper layer.
3. Gently rotate the glass rod in the liquid, at the interface
of the alcohol and detergent
mixture. Take care not to mix the
layers too much or to break up the fragile DNA. The
white web of mucus-like DNA can be
drawn from the tube with a Pasteur pipette and
resuspended in 4%
sodium chloride solution.
4. Dry the DNA thoroghly with a hair drier. IMPORTANTDont
cook the DNA. Hold your
hand near the loop as you dry.
If its too hot for you to bear, the DNA will be cooked.
After 5-10
minutes the DNA will be dry, forming a white film on the loop.
5. Pipette 100 ml
of TE buffer into a microcentrifuge
tube. Rotate the loop holding the DNA in
the buffer until DNA dissolved. The
thin liquid will become more viscous as the DNA
dissolves. The DNA can be stored
in a freezer at -20oC
at this stage, and keep until it is
needed.
Loading the gel.
1. Add 10
ml of bromophenol
blue loading dye with added SDS to
the 100 ml of
DNA
solution.The
SDS in the loading dye helps to degrade proteins that may remain in the
nucleic acid preparation.
2. Mix the DNA solution with the loading
dye thoroghly, using the microsyringe.
Load ~20 ml
of the liquid into a well.
3. Run the gel as usual.
Safety
This protocol presents no particular safety hazards, although
care should be exercised when chopping the onion or using a liquidizer.
IMPORTANT NOTE
Nucleic acids prepared in this way will not be
very pure. The essential purpose of the technique described
is to demonstrate the major principles involved in the extraction of
DNA from tissue.
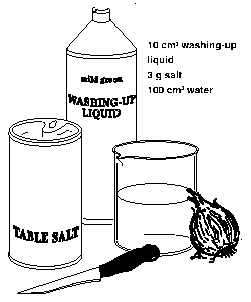
10 cm3 washing-up
liquid 3 g salt
100 cm3 water.
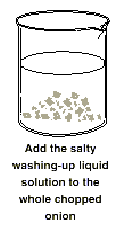
Add the salty washing-up liquid
solution to the whole chopped onion

Place in water bath at 60oC
for 15 minutes

Cool the mixture by standing it in jug of ice for a few
minutes
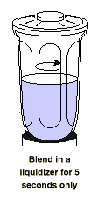
Blend in a liquidizer for
5 seconds only
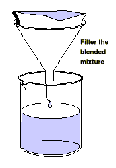
Filter the blended mixture

Add 2~3 drops of protease
enzyme to 6 cm3 of onion
extract
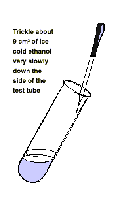
Trickle about 9 cm of ice
cold ethanol very slowly down the side of the test tube
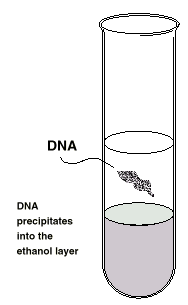
DNA precipitates into the ethanol
layer